:max_bytes(150000):strip_icc()/GettyImages-55992177-841c6b60242e426a92881ddb980020ff.jpg)
(Note: Below mentioned radii are the van der Waals radius in picometer (pm)). How to calculate the atomic radius of an element?Ītomic radius of all the elements are mentioned in the chart below. As you go down a group, new electron shells are occupied which extend further from the nucleus, increasing the atomic radius. I think the point of this question is for you to realise that options 1 and 2 can’t be the correct answer. In Fe, Co, Ni & Cu both the effects nearly balance each other keeping the atomic radius same. Why are atomic radius of Fe, CO and cu increasing? The atomic radii of the elements of 3d transition series are as under. The atomic size from Cu to Zn slightly increases because shielding effect is more than effective nuclear charge due to d10 structure of Cu and Zn. The atomic size from the Fe to Ni almost remains the same because both the effects balance each other. Why does the atomic size remain the same from Fe to Ni? This is due to the extra screening by the 3d electrons which offset the increasing pull by the additional protons going from Fe to Co and Ni on the outer 4s electrons. Which group has nearly same atomic radius?Īnswer: Fe, Co and Ni are transition metals in the same period where the atomic radii do not vary much. Does cobalt or nickel have a larger atomic radius?Ĭations are smaller than the corresponding neutral atoms, since the valence electrons, which are furthest away from the nucleus, are lost….Periodic Trends - Atomic and Ionic Radii. In zinc all the 10 d electrons are available which provide very large screening effect and decreases the effect of nuclear charge on the electrons which results in repulsion in the electrons of zinc which ultimately lead to large atomic radius. Why does zinc have a larger atomic radius? 
Which elements have the same atomic radii?įe, Co, Ni. However, even for atoms of the same type, atomic radii can differ, depending on the oxidation state, the type of bonding and – especially important in crystals – the local coordination environment. Atomic NumberĪre the atomic radii of all atoms the same? The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius….Empirical Atomic Radii. Why does manganese have a larger atomic radius? 3) So, in Fe and Ni both these effects nearly balance each other keeping the atomic radius almost same. Why do Fe and Mn have the same atomic radius?ġ) Atomic radii is inversely proportional to the effective nuclear charge, and as we move left to right across a period, the nuclear charge increases. 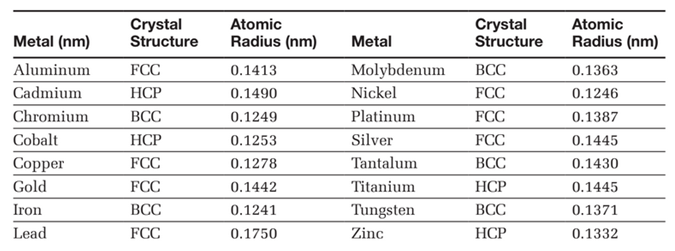
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
